An oxygen atom shares an electron pair with each H atom. The answer is option A.
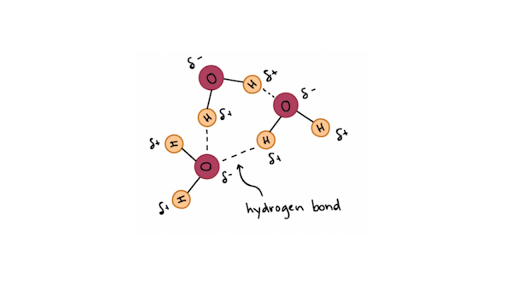
Hydrogen Bonds In Water Article Khan Academy
When there were 4 innings left the team was losing by a score of 17 to 6 points.

. Two hydrogen atoms each share their 1 electron. Chemistry questions and answers. Therefore an oxygen atom combines with two hydrogen atoms by sharing of electrons.
Thus we can conclude that the statement an oxygen atom shares an electron pair with each H atom best describes the bonding in a water molecule. There are two pairs of electrons on the central oxygen. Which of these best describes the bonding between oxygen and hydrogen in the water molecule.
Elements are pure substances that are made up of only one type of atom and. Polar bonds and non-polar bonds Non-polar bonds and non-polar molecule Polar bonds but symmetry Bent molecule shape with tetrahedral electron clouds 2. Hydrogen can only form 1 bond.
Choice C A metallic bond will form between the hydrogens in each water molecule. Since oxygen is more electronegative as compared to hydrogen atoms the shared electrons are attracted towards the oxygen atom. And make a water molecule H 2 O.
_____ bonding is a strong bond that occurs between the hydrogen end of a molecule and the fluorine oxygen or nitrogen end of similar molecules. Which statement best describes a covalent bond. A diatomic molecule shares three pairs of electrons.
Kims softball team was playing in the championship game. A water molecule consists of one oxygen atom bonded to two hydrogen atoms by covalent bonds. Consider the molecule shape and bonds in the molecule CCI4.
Only two of the six outer-shell electrons of oxygen are used for this purpose leaving four electrons which are organized into two non-bonding pairs. Choice D A covalent bond will form between the hydrogen of one water molecule and the oxygen of the other water molecule. A bond formed by sharing of electrons is known as a covalent bond.
It is rather covalent bonding that takes place between O and H. A bond formed by sharing of electrons is known as a covalent bond. By sharing the two electrons where the shells touch.
An _____ is the smallest particle of a covalent compound that still has the properties of the compound. This is a picture of a water molecule. Therefore an oxygen atom combines with two hydrogen atoms by sharing of electrons.
What best describes the bonding in a water molecule. In a covalent bond electrons are shared between atoms. Cohesion is the bonding of the same molecule such as water to water while adhesion is bonding to different kinds of molecules How.
Q8 The molecules that contains N-H O-H or F-H bonds can form hydrogen bonds which is the strong intermolecular attraction force. The electrons are thus not transferred. 2In both structures the most abundant noncovalent interactions are the multiple hydrophobic interactions between the.
In a covalent bond electrons are shared between atoms. Which statement best describes hydrogen bonding. With oxygen to form two covalent bonds.
Your body the water you drink and even the air you breathe is filled with atoms. Each hydrogen atom can count 2 electrons in its outer shell. Everything in the universe is made of tiny particles called atoms.
Hydrogen The _____ of a solute is defined as the concentration that is reached in a saturated solution at a. The answer is option C. Electrons are shared between two atoms here O and H Which allow both.
1 The electronegative hydrogen atom in one molecule and an electropositive atom in another molecule experience attraction. In the last 4 innings her team scored t. The electronegative hydrogen atom in one molecule and an.
The answer is option B. Which of the following best describes the water molecule. A water molecule is formed when two atoms of hydrogen bond covalently with an atom of oxygen.
The bonding in water molecule is not similar to ionic bonding. A chemical bond that involves sharing a pair of electrons between atoms in a molecule. In water each hydrogen nucleus is bound to the central oxygen atom by a pair of electrons that are shared between them.
A hydrogen bond will form between the hydrogen atom of one water molecule and the oxygen atom of the other water molecule. Thus we can conclude that the statement an oxygen atom shares an electron pair with each H atom best describes the bonding in a water molecule. Atoms are the basic building blocks of the universe and the same blocks build many different things.
Q7 Water molecule has 2 bonds and 2 lone pairs around the central atom oxygen therefore its molecular shape is bent. Questions in other subjects.
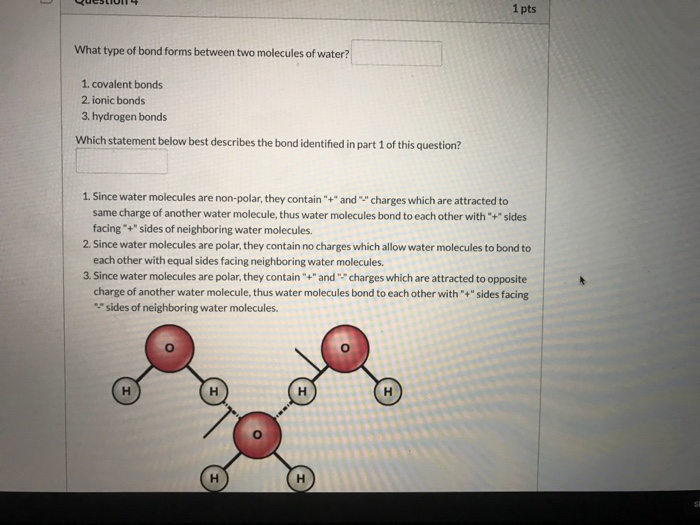
Solved 1 Pts What Type Of Bond Forms Between Two Molecules Chegg Com
What Type Of Bond Attracts One Water Molecule To Another Quora

Solved Question 6 Which Of The Following Best Describes Chegg Com
0 Comments